Your cart is currently empty!
Author: Michael Martin
-
Tools & Equipment
Why you clean How you clean…Part 10
It’s All About the Cost of Labor, NOT the Equipment
Listen to the Audio Version:
0:000:00
The Cost of Labor:
Invariably, Cleaning costs are: 90% labor, 5% consumable materials, 5% the amortization of the equipment used. Selecting ‘cheap‘ tools and equipment on cost over production value NEVER pays off. No decisions concerning the purchase of these durable goods should ever be made in an effort to “pinch pennies”. Saving 10% on equipment that adds 10% to your cleaning time will always cost you 9.5% more in total cost to clean. Is this clear? If you skimp on the quality of your tools, and it adds more labor costs to your program, then you have clearly made a bad decision.
The Value of Sustainability:
The same may be said for floor care products – with the numbers working out almost identically. Choosing a floor coating and care system that forces more deep scrubs, more strip outs or more burnishings will never create the sort of savings that matters in the long run. Select systems, tools, equipment and products that cut your labor – for the inevitable will forever arise – and you will be so much happier with your budget value.
Let me give you some numbers. If you choose a finish that will not repel soils, and must be stripped off annually, then you have just added 300% to your floor care labor costs over a program that can be restored with scrub-recoating or scrub-burnishing for a minimum of three years.

The Proof in Validation?
Every point we have made here can be put to the test, both before you buy and during the course of your fiscal cycle. If you need assistance in analyzing these validations – or presenting them to your ‘bean-counters’ please don’t hesitate to ask. You’ll both be happy with the data.
-
Antimicrobial Coatings
Cleaning Basics…Part 11
Why Antimicrobial Protection is Essential
Listen to the Audio Version:
0:000:00
This frantic preoccupation with disinfection is illogical.
Think about it. ‘Registered’ disinfectants are tested and certified microbial pesticides, meaning that they are proven to have the capacity to kill bacteria, fungi and viruses within a given period of time – usually within 10 minutes. Many of these pesticide labels insist that after that period they be rinsed with potable water. Do you know why? It is because they contain toxic or at least harmful ingredients that must be rinsed away or they will be detrimental to the occupants of the spaces in which they have been used.
These classes of such disinfectants are called quaternaries and phenolics, and they still account for too high a percentage of all disinfectants used. Cleaners and housekeepers don’t have the time to wait the prescribed period to remove the residue, so there it sits, on the tables where children eat their lunches, on the toys toddlers put in their mouths and on the surfaces so many people touch ahead of rubbing their eyes, wiping their mouth or scratching their nose. The industry is poisoning the population.
The other common class of disinfectants are called oxidizers, and they are not nearly as toxic or hazardous, and in most cases these do not leave any residue, but they also have no lasting effect. These are the peroxides, hypochlorous acids and chlorine dioxide products.
So, let’s crunch some numbers…
Let’s say that the first two classes are used as prescribed: applied and removed after 10 minutes with clear, clean water. Or the oxidizer is applied, and requiring no rinsing burns off into the air and is gone. Either way you have a window of purity, an assurance of sanitization of just 10 minutes.
The first infected person to touch, breathe or otherwise deposit bacteria on the previously disinfected surface will ruin it for the other 99.94% of the day.
What is needed is a non-toxic means of protecting the surfaces people touch around the clock and for days on end. Protecting the surfaces will protect the people, it’s really that simple. And as you will see in the segment to follow, the concept of antimicrobial protection is nothing new, with many metals having been recognized millennia ago for their germ control and anti-spoilage properties.
-
Need for Validation
Cleaning Basics…Part 13
How Do You Validate Your Cleaning Methods?
Listen to the Audio Version:
0:000:00
“Looks Good to Me!”
For centuries, the only means of validating if an area’s cleanliness was by look or smell. Even the “white glove test” does not properly speak past fine soils – dust, to the presence or absence of microbial contamination. The vast majority of cleaning programs are graded only on what data can be collected by our senses:
- Does it look good?
- Does it smell right?
- Are we hearing complaints?
New Options Abound:
Now, thanks to the study of little lightning bug, with its conversion of ATP -Adenosine TriPhosphate – into illuminating energy, there are tests that can provide empirical data as to the presence or absence of microbes on surfaces. Furthermore, there are bacteriological test plates that may be used in the field and UV lights that can show different types of soils present in restrooms.
What is Practical for You?
This is where the need for a cost-benefit analysis may be in order. So please don’t hesitate to drop us an email and we’ll help you to more effectively prove what is really “clean”!
-
Three Actions of Cleaning
Cleaning Basics…Part 3
The Three Actions of Cleaning
Listen to the Audio Version:
0:000:00
It is hard to imagine cleaning being complete without all three of these “Actions“, so let’s take them in order.
Action #1: Chemical Action
You’ve washed dishes, right? Have you noticed that dishes that have time to soak is the hot, sudsy water are easier to clean than when you don’t pre-soak them? This is due to the maximization of both the chemical (dwell-time) and mechanical (hot water) working to do the majority of the work for you. The hot water, coupled with the detergent or chemical action of the dish soap reduces your workload, your mechanical action or scrubbing of the dishes.
Action #2: Mechanical Action
Mechanical Action is usually friction. The abrasion of a scrub pad, the pressure of a brush bristle, the scrapping action of microfiber – all are mechanical “actions” that enhance cleaning performance and efficiency. The increased presence of mechanical action is directly proportional to the decreased need for chemical action. To refer back to our dishwashing analogy above, the less you soak, the more you need to scrub.

Action #3: Full ExtrAction
This third “Action” is the one most overlooked, but every bit as essential as the others. Full retrieval of wash water is essential to a “complete clean”. This is what makes string mops and mop wringer pails so horrible a tool. Yes, they deliver chemical action, and yes, they provide a measure of mechanical action; but leaving 30+% of the [dirty] water behind on the floors does not make for a clean floor. By comparison, most scrubbers will extract 97% up off the floor!
-
Use of Enzymes
Cleaning Basics…Part 7
What Are Enzymes & How Do they Help with Cleaning
Listen to the Audio Version:
0:000:00
What are Enzymes?
Enzymes exist in every corner of the Earth, and are essential to life, but most people have no idea what they are. We’d like to change that by shining some light on their role in creation.
Enzymes are proteins that, when introduced to organic materials, they act as a catalyst by jump-starting reactions without being part of the reaction. In this case they act as a catalyst of decay. Enzymes attach themselves to specific substances, like a key in a lock, and unlock the bond between the substance components.
They are the key that shuts down the security system within a compound and allows for the destruction of what holds the unit in cohesion.
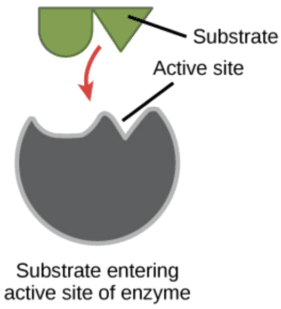
Types & Keys:
As seen above, enzymes are designed with key-sites that are specific to the organic soil type. Different substrates require different key configurations. Here is a quick breakdown of some of the types of enzymes found in cleaning compounds:
- Protease – designed to break down protein soils into peptides and soluble amino acids.
- Amylase – designed to accelerate the breakdown of starches and carbohydrates
- Lipase – designed to accelerate the breakdown of triglycerides into consumable fatty acids
- Cellulase – designed to breakdown cellulose and similar fibrous materials.
Enzymes are not produced from thin air, nor are they commonly built into detergents by themselves, they are generally coupled with benevolent bacteria.
What Happens Next?
Enzymes work hand-in-hand with specific benevolent bacteria that produce and project the enzymes. The enzymes launch the initial attack, breaking down the matter so that it can then be consumed by the bacteria. The bacteria uses the nutrients to support it self through metabolism. I like to compare it to a brick wall. The wall itself is too strong and large to consume, so the enzyme go in ahead to pulverize the mortar, making the bricks then small enough to eat. This cycle will continue until the organic soil load is diminished. In summary, enzymatic products are perfect for areas with high amounts of organic soil: seeking out, breaking down and consuming organic soils until they are gone.
-
Detergents & Microfiber
Cleaning Basics…Part 9
Using Microfiber to Reduce the Need for Detergents
Listen to the Audio Version:
0:000:00
Detergent Chemistry Review:
Generally speaking, detergents are a synthetic versions of soap. Detergents use an ionic-charged component called “surfactants” (Surface-Active-Agents) to produce a solution that reduces natural water tension, and draws water and other particles together. Sounds pretty good, right?
What is in an ion?
In this case, what ions are doing is carrying and transferring very small magnetic charges from one particle or substance to another, and once bound, they are very difficult to break away. Ionic charges may be innate to a chemical compound, or they may be produced by mechanical action – in this case – friction. Click HERE to read more on ionic surfactants.
How is Microfiber Supreme as a Cleaning Tool?
The illustration to the right demonstrates the difference between an extruded microfiber strand and that of a cotton fiber. Unfortunately, the illustration also shows them as being of equivalent size. This is not the case. A strand of microfiber is less than a third the diameter.
Microfiber is also produced of a synthetic material that creates a powerful static-electric charge as it is passed over a surface. So you can certainly visualize that as a microfiber cloth is wiped across a surface, it builds up an attracting charge AND it draws the soil and bioburden into its extruded recesses.
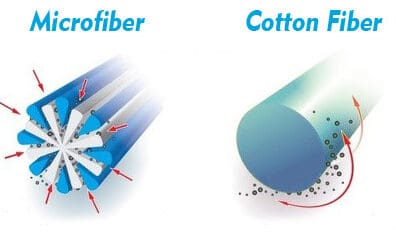
How Can Detergents Ruin Microfiber?
Now please consider the second illustration on the right. Each of these extruded fibers is in a strand, or bundle of filaments, which are then bundled again into a workable yarn containing hundreds of filaments. Each of these are stitched into a tight weave of looped ends that create a tight material for dusting or cleaning surfaces and floors.
If the wrong detergent is used with a microfiber cloth, one that adheres like glue to the extruded surfaces of the yarns, strands and filament fibers, then the material will neither be able to mechanically function to create its charge or to capture its soils.
In short, many general purpose cleaners will repel microfiber from surfaces, so that it cannot scrape soil from the surface, capture and contain it for removal; and ALL detergent-disinfectant (quaternaries) will fill the extruded surfaces with a coating of residue that will fill and kill the cloths ability to do its job.

-
Detergent Alternatives
Cleaning Basics…Part 8
A Consideration of HOCl or other Onsite Solutions
Listen to the Audio Version:
0:000:00
The Generation of On-site Generation:
In the past 10 years, there has been a veritable wave of manufacturers offering devices of various sizes that can take plain water and a touch of table salt and generate electrolytic hypochlorous acid for disinfection onsite. Depending on the power of electric consumption, and the percentage of salt added to the water solution, their claims offer some positive and some negative aspects and value. Generally what is happening with this process is the ‘splitting’ of the water molecule into water plus radical oxygen atom plus radical hydrogen atoms. The hydrogen and the oxygen atoms will then each have strong, but loose ionic charges that attract molecules with opposite charges. Please forgive the raw analogy, but the process can best be illustrated as breaking up a thousand happy homes to create wandering ex-husbands and ex-wives each looking for new mates. The salt added to the water encourages some of these unattached atoms to form a new molecule with a hydrogen atom, an oxygen atom plus an atom of chlorine from the salt. This create Hypochlorous Acid, or HOCl, a weak and unstable aqueous solution.
What is Good about These Devices:
First off, let me state that those units that are most affordable and practical for homes, daycares, schools, clinics and other businesses must be so small that they can only deliver a percentage of active HOCl to create a sanitizer. However, what they are positively providing to the market is a focus upon the “oxidizing” class of disinfectant and moving these same type of facilities away from using off-the-shelf, RTU bottles of liquid poison – quaternaries. This drives down the need for and supply of the 99% water solutions being stocked, stored and shipped as was described in another post.
What is Not-so-Good about These Devices:
As mentioned, these devices electrically convert water and salt into an oxidizing solution. But by necessity, they must also generate another byproduct in the same bottle that robs from the potential of the HOCl, holding it back and continually inviting it to revert to water and salt. In short, the sanitizing power is very short-lived, not easily verifiable, and too often – users are thinking they are sanitizing when in reality they are cleaning with just water.
The Better Solution:
We went another route in our search to eliminate water in shipping, and to create a solution that is an oxidizer which is far more verifiable and stable. The answer is ClO2, and we will explain why in future Cleaning Basics segments.
-
Role of Dwell-Time
Cleaning Basics…Part 6
What is the Role of
Dwell-Time in Cleaning
Listen to the Audio Version:
0:000:00
Detergency & Dwell-Time:
As mentioned earlier, detergents and soaps work much on the same premise of combining a vehicle (water) with a natural or synthetic surfactant which has both a water-attracting property (on one end) and a soil-attracting property (the other end). These surfactant molecules need time to move through the cleaning solution, locate the soil and attach itself to these molecules. However, beware, the same property that attracts surfactants to soil, does so also to surfaces. Detergent that is left behind, after the water dries, will continue to attract soil to surfaces.
Reducing Dwell-Time:
Surfactancy and dwell-time, when applied to surface cleaning, may be reduced and offset by friction. Have you ever noticed how quickly a power washer removes soil? Our a scrub brush reduces cleaning times? Or wondered why hot water cleans better and faster than cold water? It’s the agitation of the molecules in hot water that causes friction. Agitation, or friction, when placed in the balance, does quickly what surfactancy does over time.

Dwell-time & Disinfection:
The exception to this is in disinfection.
Laboratory testing, required of all registered disinfectants, validates the performance of biocides on a number of levels – one of which is called the ‘CT’ Factor – Concentration x Time. To achieve the appropriate efficacy of disinfectants, dwell-time, concentration and soil loads must ALL be adhered to with almost scientific precision. To learn more on this subject, read our article on ‘The Ignorance of Protocol‘ on our Thoughts page.
-
Role of Water
Cleaning Basics…Part 4
The Essential Role that Water Plays in Cleaning
Listen to the Audio Version:
0:000:00
Solvents, Surfactants & Vehicles:
In the same manner as was mentioned in our previous post – the Three Action so Cleaning, so also there is another rule. This rule states that most cleaning compounds, both soaps & detergents are made up of 3 components: a solvent to break the soil free from the surface, a surfactant to contain the soil in the vehicle, which is generally water. The water is called a vehicle because it carries the other components to the soil, and then carries the soil away from the cleaned surface.
Surface Tension of Water:
Water does not naturally spread, saturate, or even collect and contain soils. This is easily seen in a glass containing water and a few drops of oil. The two clearly keep to themselves until there is the introduction of the detergent that…
- breaks the bond of the surface of the water; and
- emulsifies the oily soil into suspension in the water.
This is a simple process to accomplish and takes only a microscopic amount of surfactant to accomplish – and it may be achieved with either dry or liquid compounds.

The Sustainable Brilliance of Water:
Water, employed as the vehicle for cleaning, is one of the most efficient exercises in sustainability possible. The world’s problem with the current state of cleaning is that far to great a percentage of cleaning products are 95% or more composed of water. This leaves millions upon millions of gallons of water sitting bottled in various container, on retail shelves, in warehouses and carried in transit. Using super concentrates (see our Synergy SB solutions line) or chlorine dioxide solutions (prepared on-site with water) to create cleaning solutions (see Envirotab), allow for water to be used, evaporated and returned to the local supply without hindrance.
-
Detergent Composition
Cleaning Basics…Part 5
What is a Detergent? and
What is a Surfactant?
Listen to the Audio Version:
0:000:00
Soaps versus Detergents:
Soaps, general composed from the fats, oils and lye (or wood ash) have been around for thousands of years. Detergents, made of synthetic ingredients were born out of wartime necessity and have barely pass their centennial birthday.
Synthetic Surfactants:
While simplistically put, detergents have 3 basic components: Surfactants, Solvents & a Vehicle (generally water). The lion’s share of the workload is carried on by the synthetic surfactant. These key ingredients have ends that are repellent opposites of one another. One end is hydrophilic (water-loving) and the other is hydrophobic (soil-attracting). This core component both allows the vehicle (water) to spread more evenly, and to convert the water to a soil-carrying vehicle. Solvents (and other lesser ingredients) are placed within these formulas to help to break the bond of the soils from the surfaces.

Other (& More Sustainable) Alternatives:
There are, however, some real downsides to using synthetic surfactants. First, most are petroleum derivatives – not renewably sourced and not readily biodegradable. On the other hand, some surfactants have been derived from plants and hence thoroughly biodegradable. In addition to these alternatives, the employment of microfiber for hard surface cleaning can eliminate the need for any chemical surfactant and the use of chlorine dioxide (for hard surface cleaning) can usually eliminate the need for any other solvent. Beyond detergent residue, biofilm (the scum produced bacteria) is the next main cause for soil build-up on surfaces – and NOTHING breaks through biofilm better than aqueous chlorine dioxide. For more information, see our page, “A Better Way to Clean Hard Surfaces“.