Your cart is currently empty!
Author: Michael Martin
-
Accolades for Chlorine Dioxide
Accolades for Chlorine Dioxide:
Chlorine Dioxide – Approved for Water Treatment:
All around the world, chlorine dioxide has been approved and recommended for the treatment and purification of water for consumption by humans, animals and plants. Among these are:





Recommended for the improvement of IAQ:
On many occasions, chlorine dioxide has been tested and recommended for the reduction of airborne pathogen load in air. Among these agencies is:





Studied and Approved for Surface Disinfection:
And of course, the tests conducted using ClO2 for disinfection are almost endless. Some of the most notable are these respected institutions:






More Articles & Information:
-
Selectivity of ClO2
Chlorine Dioxide Basics…Part 4
An Introduction to the Unique Characteristics of Chlorine Dioxide
Disinfection through Oxidation:
Atoms are composed of matter with electrically charged particles: Protons (+), Electrons (-) and Neutrons (0). The class of biocides referred to as “oxidizers” all work by means of an electron exchange – one atom gains and the other atom loses an electron. All oxidizers may be measured by means of their ‘oxidation strength’ and their ‘oxidation capacity’ or ‘potential’. The higher the ‘strength’ the more powerful the attack, the higher the ‘capacity’ the greater the longevity. It can be compared to two armies – one with a single large cannon (but just a few cannon balls), and another with several small artillery AND an abundance of ammunition.
ClO2 – Compared to Other Oxidizers:
Let’s use these two classifications to make a comparison:
Oxidant Strength Capacity
Ozone 2.07 2 e-
Peroxide 1.78 2 e-
Bleach 1.49 2 e-
Bromide 1.33 2 e-
ClO2 0.95 5 e-
What this means is that of the five compounds in the oxidation class, chlorine dioxide has the lowest capacity to do harm to humans and the highest capacity to do harm to microbial metabolism. From this chart you can see that ozone and peroxide can do twice the damage, but with only a third of the capacity to rob electrons and disarm microbes.
To use our artillery analogy, ClO2 may have the smallest gun among the oxidizers, but 250% more ammunition, targeting is easier and damage to environmental surfaces is greatly reduced.
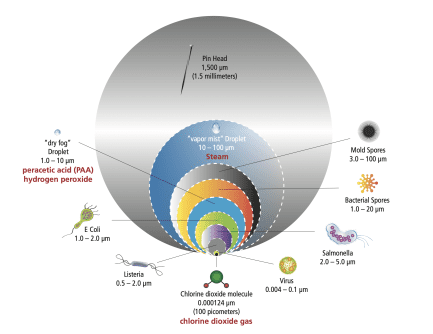
Size Matters:
The very fact that the chlorine dioxide molecule is so small is what makes it so quick and effective. When looking for matter to oxidize, the ClO2 molecule begins with the smallest opponents first – viruses, bacteria (of various sizes), spores, fungi and protozoan. It’s strength is exhausted only progressively, and not before it exacts destructive force on the toughest of microbes.
We will cover this in much greater detail with a variety of scholarly reports and laboratory and practical studies.
-
Water | Air | Surface
Chlorine Dioxide Basics…Part 5
ClO2: The Water, Air and Surface Disinfectant
Listen to the Audio Version:
0:000:00
ClO2 in Air Purification:
Unique among the oxidizing class of disinfectants, Chlorine Dioxide has been frequently and successfully used to purify the air of odor and other contaminants. Among the most famous of which was the use of it in the 1976 outbreak among the attendees of an American Legion Convention at the Bellevue Hotel in Philadelphia PA. Radical steps were taken to save the building from demolition, and the city’s inhabitants from further exposure, the hotel was enclosed in a tent and filled with ClO2 gas. The treatment was a complete success. Today, ClO2 is used for everything from localized odor control to the reduction and removal of airborne microbes from the air.
ClO2 in Water Treatment:
It is not feasible to deliver perfectly sterile water for drinking, animal hydration or plant irrigation. Trace bacteria is found in most water sources – especially well water. As the water sits idle in supply lines, bacteria reproduces, colonizes and constricts the lines with its defensive system. This increases the potential for contamination of people, animals and greenhouse-grown plants. ClO2 is unique in its ability to break down and control bacteria and biofilm at levels that are perfectly safe for drinking.

ClO2 for Surface Disinfection:
Thousands of studies have been conducted to demonstrate the superior ability of chlorine dioxide to perform surface disinfection without leaving a sticky or harmful residue behind. The science of this is simple: the water delivers the gas (ClO2), the gas destroys the contaminating microbes and anything left dissipates into the air, leaving only the water behind. We will dedicate a number of pages to present these details in our Documentation Section.
-
The Superior Oxidizer
Chlorine Dioxide Basics…Part 3
How is Chlorine Dioxide Different and Better than Bleach?
Listen to the Audio Version:
0:000:00
Bleach – The “Go To” Product:
For decades, bleach has been the “go to product” for disinfection. When deep disinfection is required, or an outbreak has occurred, facility and housekeeping managers have mandated the use of bleach for hard, inanimate, non-porous surfaces. While they are right to assume that this oxidizer has some value, it is far from being the best delivery system for chlorine.
ClO2 – The Better Choice:
The problems associated with using bleach are manifold: Bleach must be used at much higher concentrations than ClO2. Bleach will react very differently depending on the pH of the surface or the solution to which it is added*, and ClO2 is not as susceptible to decay by interaction with organic materials. To summarize – you get 5-10 times more ‘bang for your buck‘ with chlorine dioxide. If you review the charts to both the left and the right here, you will see that while both forms deliver free-available chlorine to the site, only chlorine dioxide does so efficiently.
*Bleach must be used in a very narrow pH range (5-7.5); ClO2 may be used in solutions ranging from 2-11 in pH.
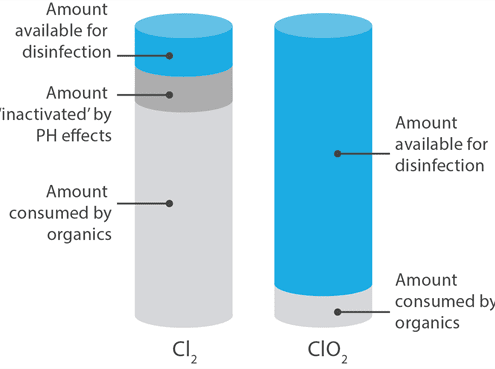
ClO2 – The Safer Solution:
For over a century it has been known that when elemental chlorine gas or liquid bleach is added to water, the acid it creates it is likely to come into contact with particular organic matter that precipitates the creation of trihalomethanes, a carcinogenic byproduct that can cause cancer in humans and animals. This never happens with chlorine dioxide, nor does ClO2 leave behind a residue of salt that is destructive to metallic environmental surfaces.
Furthermore, ClO2 is the ONLY selective oxidizer and does not naturally degrade ferrous metal derived materials like steel and chrome.
-
What is Aqueous ClO2?
Chlorine Dioxide Basics…Part 2
What is Aqueous or Waterborne Chlorine Dioxide?
Listen to the Audio Version:
0:000:00
Chlorine Dioxide – Along for the Ride:
As mentioned in our earlier post, ClO2 is a gas, and does not hydrolyze in water. This means that its characteristics do not change, its components do not break down and it does not become what we might call a traditional “solution”. Chlorine Dioxide is simply a gas, delivered by the vehicle – water – and it’s just along for the ride. Let’s continue with the analogy: put a seat in a car and it becomes an integral and essential part of the car. Put a passenger on the seat and he or she does not become an integral part – removing the passenger does not detract from the car, remove the seat and it is less than a car. In the case of chlorine dioxide, you can tell it’s there from the distinctive yellow color.
Chlorine Dioxide – Looking for a Destination:
This molecule, which can be added to water without being altered in state, function or power, is on a hunt. The ClO2 molecule, somehow, has an innate understanding of its place in Creation. It is designed and determined to find and attack pathogenic invaders. While it employs oxidation to disinfect, it is selective – looking specifically for (from smallest to largest) microbial contaminants. Being so small in size, it can simply leave its aqueous vehicle, pass through the lipid layer of a virus or the membrane of a bacteria and begin its work of dismantling the matrix of the microbe.
Generated Organically:
The generation of this molecule is closer to home than one might think.
Being reacted into the molecular union of Chlorine and Oxygen, designed to work at extremely low concentrations and being selective in its attack strategy, what is a better compound to place within our own white blood cells?
Three of the Earth’s most abundant resources are Air, Water and Salt. Without any one of these we cannot live, and furthermore, without the formation of the ClO2 – chlorine from the salt, and oxygen from air and/or water – we would have no means of fighting off infection or decay.
Delivered and Deposited:
Once the ClO2 molecule is delivered to the intended target, it is free to go about its business. To return to our analogy, once the water – let’s refer to it as the Uber – gets to the site for cleaning, deodorizing or sanitizing, the passenger exits the vehicle and begins its work.
What then begins is its selective prosecution of viruses, bacteria and fungi until it is completely exhausted of resources or it has completed its assignment. The gas then evaporates into and becomes a sanitizing component within the air.
-
What is Chlorine Dioxide?
Basics of Chlorine Dioxide…Part 1
The Most Basic Answer: What is Chlorine Dioxide?
Listen to the Audio Version:
0:000:00
Chlorine Dioxide – Discovery & History:
Chlorine Dioxide, a molecule consisting of just one chlorine and two oxygen atoms, was first discovered by Sir Humphrey Davies in 1811. For nearly 140 years, little was done to explore or employ this discovery into any form outside of the laboratory.
Then, in 1944 out of measure of some desperation, Niagara Falls, NY had an onsite chemical generator installed to address a serious problem. At issue was the odor and taste of their water after purification by means of their previous free chlorine treatment program. They changed their water treatment protocol from using hydrolyzed chlorine to using gaseous chlorine dioxide. The results were amazing. Within 30 years, another 3000 facilities had converted to this superior biocidal system for water treatment. Soon after, the Pulp & Paper industry converted their processes to include Chlorine Dioxide which also became an alternative for large scale food processing sanitation.
Molecular Characteristics:
Chlorine Dioxide is a gas, and remains such at all temperatures over 51 degrees Fahrenheit. Even when added to water, it does not hydrolyze – but remains a gas. It is 10 times more soluble in water than straight chlorine. This means that may be effectively delivered by water, but it is far more stable and effective than other forms of disinfectant. We will speak more to this later.
“The Perfect Biocide”:
In the beginning, ClO2 could only be either generated onsite with massive equipment and hazardous chemicals or delivered in bulk totes. This was an expensive and dangerous proposition in 3000ppm and higher concentrations. Now, be it through onsite generation through liquids, powders or tablets, the majority of the safety concerns have been addressed. Furthermore, being the ONLY singular chemistry able to sanitize WATER, AIR and SURFACES, the World Health Organization has deemed it the “Perfect Biocide”.
-
Small Volume Calculations (Metric)
Calculating ClO2 for Small Volumes Solutions – Metric
Please note: While tablet volumes may be shown in decimal point quantities, the tablets are NOT to be cut. Simply round the quantity up or down as required.
-
Small Volume Calculations
Calculating ClO2 for Small Volumes Solutions – U.S.
Please note: While tablet volumes may be shown in decimal point quantities, the tablets are NOT to be cut. Simply round the quantity up or down as required.
-
Open Volume ClO2 Calculator
Open Volume Calculations for ClO2 Concentration
Please note: While tablet volumes may be shown in decimal point quantities, the tablets are NOT to be cut. Simply round the quantity up or down as required.