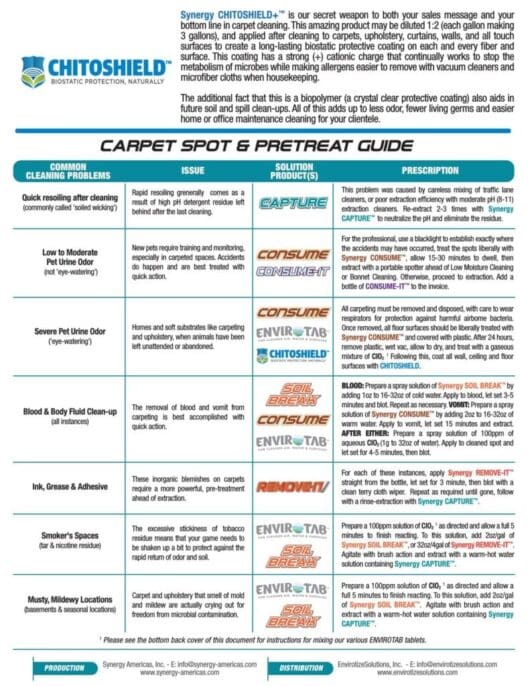
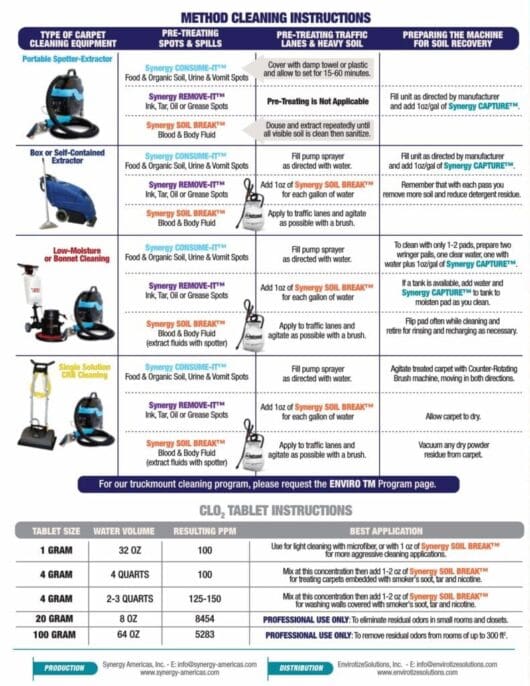
Stay Connected...Drop Us a Line
For best user-experience, hit 'TAB' to move from field to field.
Chlorine Dioxide Basics...Part 3
How is Chlorine Dioxide Different and Better than Bleach?
Listen to the Audio Version:
Bleach - The "Go To" Product:
For decades, bleach has been the "go to product" for disinfection. When deep disinfection is required, or an outbreak has occurred, facility and housekeeping managers have mandated the use of bleach for hard, inanimate, non-porous surfaces. While they are right to assume that this oxidizer has some value, it is far from being the best delivery system for chlorine.
ClO2 - The Better Choice:
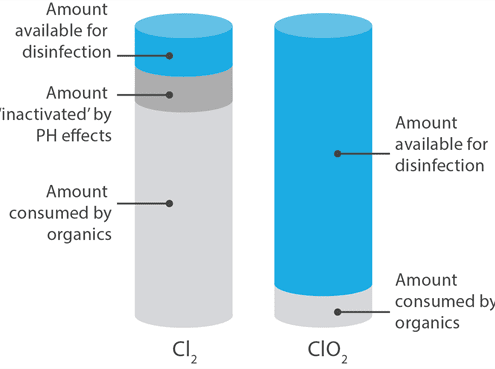
The problems associated with using bleach are manifold: Bleach must be used at much higher concentrations than ClO2. Bleach will react very differently depending on the pH of the surface or the solution to which it is added*, and ClO2 is not as susceptible to decay by interaction with organic materials. To summarize - you get 5-10 times more 'bang for your buck' with chlorine dioxide. If you review the charts to both the left and the right here, you will see that while both forms deliver free-available chlorine to the site, only chlorine dioxide does so efficiently.
*Bleach must be used in a very narrow pH range (5-7.5); ClO2 may be used in solutions ranging from 2-11 in pH.
ClO2 - The Safer Solution:
For over a century it has been known that when elemental chlorine gas or liquid bleach is added to water, the acid it creates it is likely to come into contact with particular organic matter that precipitates the creation of trihalomethanes, a carcinogenic byproduct that can cause cancer in humans and animals. This never happens with chlorine dioxide, nor does ClO2 leave behind a residue of salt that is destructive to metallic environmental surfaces.
Furthermore, ClO2 is the ONLY selective oxidizer and does not naturally degrade ferrous metal derived materials like steel and chrome.